The anode material in almost all of today’s secondary lithium-ion batteries is graphite. In order to boost the energy density, we would love to replace the graphite by lithium metal. Many people work on this goal. Unfortunately, the lithium metal anode tends to form dendrites during electrochemical plating, eventually ending the battery life by an internal short-circuit.
In our previous application note “Lithium metal dendrites: Pictures speak louder than words” we have presented a time-lapse video showing the lithium dendrite growth in a lithium metal battery using a graphite strip as the working electrode, and lithium metal as the counter electrode. We still own you the proof, that there are no dendrites when using a non-metal lithium source as the counter electrode. Here you go!
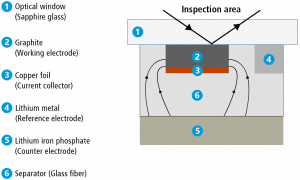
With our ECC Opto-Std test cell, we placed a graphite strip next to a piece of lithium metal foil under the sapphire window. A glass fibre separator and a disc-shaped lithium iron phosphate (LFP) electrode were pressed against this arrangement from below. The graphite strip was connected to the working electrode of the potentiostat, the LFP electrode to the counter electrode and the lithium metal to the reference electrode. In this way, the lithium metal was used as a reference electrode, and lithium ions were only moved back and forth between the LFP and graphite electrodes. The following video shows how the lithiation / delithiation progresses along the width of the graphite electrode during the electrochemical cycle.
Lessons learned:
Using an additional reference electrode, the ECC-Opto-Std test cell allows visualization of the potential and Li-ion concentration gradient in lithium-ion anodes and cathodes. With a non-metallic counter electrode the life time of the set-up is no longer limited by dendrite growth. Li-ion is the answer!
You may complain that, with a light microscope, we don’t see what is going on in the LFP cathode. Fortunately, the ECC-Opto-Std can easily be adapted to other techniques such as Raman or X-ray.



Comments are closed.