Frequently asked questions
General
Can I order directly from EL-CELL, or do I need to contact a distributor?
We generally offer our products and services worldwide, either directly or through one of our distributors. In any case, you can send your order by e-mail to [email protected] or use our contact form. We will contact you or forward your request to the respective distributor in your area. You can also order via our webshop, which offers consumables and spare parts. If a distributor is responsible for your country, you can send them a request via the webshop.
What means of payment do you offer?
We offer payment by credit card (MasterCard and VISA), paypal or bank transfer.
I would like to pay by credit card from outside the EU. What do I need to consider?
You can use MasterCard or VISA for payment. Please make sure that your card is enabled for EURO as currency and that the invoice amount does not exceed the limit of your card.
How can I report a technical problem with one of your devices?
If you have a technical problem or a question about the use of one of our products, please do not hesitate to contact us. Our experienced electrochemists will be happy to help you with any application questions. In order to help you quickly in case of a defect, just send us a detailed description of your problem, and if possible, pictures of the defective parts. We will get back to you with a solution as soon as possible. The necessary steps to return a product are listed here.
Products
Can I borrow your potentiostats for testing?
Yes, a rental of our PAT-Tester-i-16 or PAT-Tester-x-8 potentiostats is possible on request, just contact us and we will provide you with further details. In addition, we offer to demonstrate our other products at your site or in our battery laboratory in Hamburg at any time.
What do the abbreviations PAT, ECC and ECD stand for?
We offer different product series, each of which can be identified by the abbreviation in its name:
PAT stands for PArallel Testing and identifies our innovative, cableless cell concept, which is particularly suitable for high throughput testing due to its easy handling. In addition to cells, the PAT series also includes the corresponding docking stations and potentiostats.
ECC means Electrochemical Cell and is used for our optical cells and other cell designs like the ECC-Ref, which are connected directly to battery testers and potentiostats by cable.
ECD stands for Electrochemical Dilatometers and characterizes all special cells that can measure the expansion of the electrodes in the nanometer range.
How can I identify whether my PAT-Cell is suitable for use with metal seals?
The compatible cell lid for metal seals (Item name: Screw cap insulated (PAT)) can be recognized by two features. It has two holes on the top and a white or black inset made of PPS in the lid, which serves as insulation.
As the two holes can also be found on older cell lids without insulation, you should always check whether the inset is present. Non-compatible cell lids have no inset at all.
Can I connect PAT-Cells respectively the PAT docking stations to any battery tester or potentiostat?
Yes, our PAT series test cells can be operated on any common battery tester or potentiostat using a PAT docking station. Most docking stations are connected via conventional 4 mm banana plugs. The PAT-Stand-16 and PAT-Chamber-16 are offered with fixed open-ended cables. Cell cables with custom connectors to connect these units to a specific battery tester are available on request at extra cost.
What is the spring force applied to the cell stack inside a PAT-Cell?
The force applied to the cell stack of the PAT-Cell is mainly determined by the disc spring attached to the lid. This force is 40 N +-5N, provided the upper electrode is between 0 and 0.8 mm thick. The electrode thickness has no significant influence on the force within this range.
This information is valid for all test cells that use the PAT-Core, except for test cells with variable force adjustment (PAT-Cell-Force).
Can I use the PAT-Cells for experiments with aqueous electrolyte?
Yes, this is possible. The cell bases of all current cells of the PAT series, except the PAT-Cell-Twin-Ref, are made of corrosion-resistant steel (1.4404) and are therefore generally suitable for use with aqueous electrolytes. Please note that this applies to the PAT-Cell only for models manufactured from September 2019 (revision no.4) and PAT-Cell-Press manufactured from June 2019 (revision no. 3).
How much electrolyte should be used in a cell?
Keep the cell “thirsty”. Just add the amount of electrolyte required to fully soak the porous components of the electrode stack – separator and electrodes, not much more. 100 µl is a good starting point and will work well with most LiB configurations. A large excess of electrolyte may cause contamination of the cell bottom and the reference pin. As a consequence, the reed contact of the insulation sleeve may lose contact, which in turn may result in scatter of the half cell voltages VxR.
Which separator to use?
We consider glass fiber Whatman (GF/A 260 µm thick) as best for general purpose use because of its superior wettability. Next best is the 2-layer-separator FS-5P made of a PP fiber layer (180 µm) and a highly porous UHMW-PE microporous membrane (40 µm). The FS-5P separator is glass-free, but only second-best in terms of wettability, reproducibility and impedance artefacts. Technical polyolefine (Celgard-like) separators are possible, but often show wettability issues. Don’t use them for single-electrode impedance measurements.
Finger type reference, which separator?
Finger type reference electrodes should only be used with glass fiber separators. We have good experience with Whatman GF/A 260 µm thick. Celgard type separators are not working well. Gas bubbles easily get enclosed between the two separator layers especially along the finger and cause artefacts due to current inhomogeneities.
Re-use (PEEK) variant of insulation sleeve: Can the standard reed contacts be re-used?
Yes, possible. However, best results with new reed contacts. If re-using: Clean with detergent wash, rinse with water, dry with compressed air, don’t touch the contact area at the reed contact, don’t bend too much. A little ultrasonic cleaning may help, but be cautious. We have seen the polyimide coating peeling off in case of excessive ultrasonic treatment.
Is it possible to use insulation sleeves in aqueous media?
Both the single-use variant (made of PP) and the re-use variant (PEEK) of the insulation sleeve can be used with many aqueous electrolytes. Just make sure that all components – sleeve, reed contact and separator – are compatible with the used electrolyte. The least problems are with neutral and alkaline electrolytes (such as KOH), while strongly acidic electrolytes (such as H2SO4) are often not compatible.
Separators:
Glass fiber (such as Whatman GF/A) or polypropylene fiber separator (such as Freudenberg FS2226) is compatible with many aqueous electrolytes. Note that the two-layer FS5P separator does not work because of the PE membrane layer; it cannot be wetted with aqueous electrolytes. For the same reason, Celgard-like separators do not work.
Reference material:
In most neutral electrolytes, activated carbon (either free-standing or coated on stainless steel 1.4404) works well as a pseudo reference material. Activated carbon is known to corrode in strongly alkaline electrolytes (such as potassium hydroxide) and so cannot be used here.
What is the right plunger height?
The proper plunger size depends on the thickness and compressibility of the lower electrode and the built-in separator. The size is given as a number at the bottom of the lower plunger. The thicker the lower electrode and the separator are, the larger the number must be. For calculation, please use our plunger size calculator.
We offer plunger sizes only in steps of 50 µm. We have found that in-between sizes do not systematically improve behaviour. The right plunger size is especially important for single electrode impedance measurements, while it is much less critical for full cell impedance measurements and constant current cycling. It is generally acceptable to be +-50 µm off the calculated value even for single-electrode impedance measurements.
We have visualized the different scenarios in a video.
How thick may the upper electrode be?
The upper electrode can be operated with a standard upper plunger up to a thickness of 800 micrometres. For thicknesses above this, the upper plunger must be adapted. Contact us for a customised solution in this case.
Single electrode impedance measurements – How to minimize artefacts?
Single electrode EIS measurements are especially prone to artefacts due to current inhomogeneities inside the electrode stack and imperfect placement of the reference electrode. Such artefacts cannot be completely eliminated, but can be minimized by the following measures.
- Use insulation sleeves with thick (260 µm) glass fiber separator (rather than separators with only some 10 µm thickness)
- Use two additional 18 mm glass fiber separators, one below and the second above the built-in separator.
- Use the proper lower plunger size.
- Observe the different quality of the two half-cell impedances. The larger of the two impedances is relatively less distorted. Note that the ratio Z1/Z2 is a function of both SOC and frequency.
Disposal of lithium rings / insulation sleeves with lithium ring
The Li metal ring is actually a stainless steel ring coated with about 1 mg of lithium metal. Just 1 mg. After use, the ring (or the sleeve with built-in ring) can just be disposed of in a beaker and allowed to react in the ambient air.
Standard test cells
How can i minimize excessive noise in half cell voltages?
Excessive noise in the half cell voltages VxR is due to sometimes poor contact between the short pin in the cell base of the PAT-Cell and the contact pad at the reed contact of the insulation sleeve. To minimize this problem
- Don’t touch the two contact points with your fingers/gloves during cell assembly
- Don’t use too much electrolyte. 100 µl is enough in most cases.
- If dirty, gently clean the short pin inside the cell base, e.g., with a Dremel rotary tool with brass brushes at lowest speed. Clean further with cotton swabs and acetone.
- It is best to use a new reed contact for each test. If reusing, clean them carefully beforehand.
Caution: The pin inside the cell base is plated with nickel. Don’t use abrasive tools as this can damage the nickel plating
Gas analysis test cells
Which cell is right? PAT-Cell-Press or PAT-Cell-Gas?
Do you always need a PAT-Press-Box to operate the PAT-Cell-Press?
The PAT-Press-Box is required to read and record the analog pressure signal of a PAT series test cell.
It is required in the following docking stations:
The following devices provide the necessary functions themselves, therefore no PAT-Press-Box is required:
Can the PAT-Cell-Gas be operated with a PAT battery tester?
Yes, this is the easiest way to operate the cell. Simply plug the test cell into a PAT-Channel of the PAT-Tester-x and connect the gas in- and outlet. The PAT-Channel can be placed inside a temperature chamber at 0 to +40°C. For more extreme temperatures, the test cell can be plugged into the PAT-Stand-1 which is connected by a cable to a PAT-Channel outside the temperature chamber. Note that the PAT-Cell-Gas only fits into the PAT-Tester-i-16 when disconnected from gas supply.
Can the PAT-Cell-Gas be operated with a third-party potentiostat?
Yes, plug the test cell into the PAT-Stand-1 and connect the stand to the cell cable of your potentiostat. For reading the pressure signal (only PAT-Cell-Gas variants P and SP), the PAT-Press-Box is required. Insert the PAT-Press-Box between PAT-Stand-1 and potentiostat or battery tester.
Which cell is right? PAT-Cell-Press or PAT-Cell-Gas?
How do i set up the PAT-Core for gas analysis measurements in the PAT-Cell-Gas?
The configuration of the PAT-Core for gas analysis is identical to the common setup in the PAT-Cell, with two exceptions:
- For the lower plunger, use the version with flow field or with perforated plate, depending on your test case.
- Use an electrode that is supported by a gas permeable current collector as lower electrode. This permeable current collector can be a mesh or expanded metal. In the manual, we refer to this electrode as “gas diffusion electrode”.
With the permeable current collector, gases generated at the electrodes can be carried out of the cell along the spiral flow field with the carrier gas supplied from outside. The term “gas diffusion electrode” is intended only to express that the lower electrode must be somewhat permeable to gas. It must not have a foil-like current collector that is impermeable to gas. Note that gases formed at the upper electrode will also enter the gas stream to some extent. It may therefore be useful to use LFP as the upper electrode instead of lithium metal, for example.
Can the PAT-Cell-Gas used with other than standard LiB electrolytes?
Yes, provided that the used electrolyte is compatible with all cell parts that can get in contact with the electrolyte. We guarantee compatibility with all common aprotic battery chemistries/electrolytes. For all other battery chemistries/electrolytes, the user must find out for himself whether the components installed in the PAT-Cell are compatible. We can only provide some general guidance.
- For the PAT-Cell-Gas, current collectors and other metallic components are made of stainless steel 1.4404 (316L). Stainless steel 1.4404 is stable in many neutral and alkaline electrolytes. It is however unstable in strongly acidic electrolytes such as sulfuric acid, and in the presence of halogenides such as NaCl.
Optical test cells
Can I use the provided borosilicate glass window with any lithium battery electrodes?
Borosilicate glass is a good choice for all common cathode materials and also lithium titanate as the working electrode. However, some anode material such as lithium metal and lithiated graphite may react with the glass window forming greyish spots of elemental silicon on the glass surface. We therefore recommend using sapphire (Al2O3) windows with anode materials.
What is the difference between the ECC-Opto-Std and ECC-Opto-Aqu?
The basic difference is that for the ECC-Opto-AQU gold and PEEK are used for the parts coming into contact with the electrolyte. Gold is stable in most aqueous electrolytes (an important exception are solutions containing chloride), but is sometimes not stable in aprotic electrolytes (gold may form alloys with Li when used as a current collector for the negative electrode in Li+ containing solutions). We have also seen gold corroding in aprotic surpercapacitor electrolytes, R4NBF4 in MeCN, when used as the positive current collector.
Please note that the PEEK polymer used in both cells for the cell body has some stability limits. PEEK is not recommended for aqueous H2SO4 at concentrations >50% (as PEEK is getting sulfonated) and concentrated HNO3. In general, using one and the same cell for both aqueous and aprotic electrolytes may cause trouble because of cross contamination, especially from the aqueous towards the aprotic systems.
Electrochemical dilatometer
What are the differences between the built-in sensors of the ECD-3 and ECD-3-nano?
The sensors of ECD-3 and ECD-3-nano are both able to detect very small expansions of the electrodes. The main difference between the two devices lies in the detection resolution. The ECD-3 uses an LVDT sensor with a resolution of 50 nanometers with a maximum displacement range of 500 μm. The ECD-3-nano uses a capacitive sensor system with a resolution of 5 nanometers and a maximum displacement range of 250 μm.
How can I clean the T-Frit? How much does a stained T-frit affect future results?
The frit is made of borosilicate 3.3 glass (Duran®) and so can be cleaned with any agents lab glass is compatible with, including e.g. hot aqueous HNO3. According to our experience, the staining of the T-frit does not affect electrochemical results.
Why is it possible to measure displacement values below zero?
The dilatometer does only measure the displacement (=change in thickness), but not the total thickness. The initial displacement reading is arbitrary and can be changed by just turning the micrometer screw at the sensor head. In practice, you turn the micrometer screw so that the initial reading is in the range of zero +/- 20 µm. After the measurement, for convenience, you substract some offset value, so as to set the displacement value exactly to zero at that point in time, where you have started the electrochemical cycle.
Potentiostats
Can the PAT-Tester-i-16 also be used to test other cell formats that do not belong to the PAT series?
The PAT-Tester-i-16 supports other battery test cells and is not just limited to the PAT series test cells. These are connected via an adapter plugged into the PAT socket. Currently, both 2- and 3-electrode coin cells can be connected via Coin-to-PAT-Adapter and 3E-Coin-PAT-Adapter. For other cell formats like small pouch cells or T-cells cells, we offer the Uni-to-PAT-Adapter and the Cable-to-PAT-Adapter.
Can test cells other than PAT-Cells be connected to the PAT-Tester-x?
The PAT-Tester-x can be used with test cells of the PAT series as well as with other test cells from EL-Cell or other manufacturers. These are connected to the Sub-D connector of the PAT channel via a special cell cable.
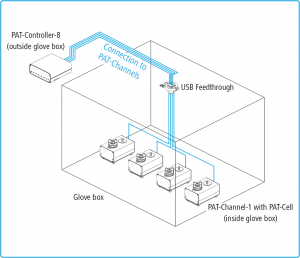
Can the PAT-Tester-x be used to cycle test cells directly in the glovebox?
Tools
Can you change the cutting tools of the EL-Cut?
No. The cutter of the EL-Cut must be precisely adjusted at the factory in order to achieve its high-precision cutting results. It is therefore permanently installed in the housing and cannot be changed.
Can I use the ECC-LiPunch to punch lithium disks that are on a carrier material, such as copper?
No, the ECC-LiPunch is only suited for punching disks of pure lithium without a carrier material.